Basic Sciences
Investigating the mechanism of prolotherapy: the fibroblast response to dextrose in vitro
Friday, March 31, 2023
12:00 PM East Coast USA Time
- SF
Sarah Fox, OMS
Campbell University School of Osteopathic Medicine
Lillington, North Carolina, United States
Presenting Author(s)
Background and Hypothesis: Dextrose prolotherapy injections represent a promising and effective, non-surgical treatment option for patients with osteoarthritis. However, the mechanism of dextrose prolotherapy is not well understood, and more studies are needed on determining the optimal dextrose concentration for therapeutic injections. Therefore, we hypothesize that hypertonic solutions of 10-25% dextrose will incite an initially damaging, inflammatory response followed by the production of growth factors and enhanced fibroblast proliferation.
Methods: The XTT assay was used to determine cell viability on MRC-5 fibroblasts directly treated with a range of dextrose concentrations (5 -25%) in complete media across 15 min-12 hr. Additionally, we investigated how the response of dextrose-exposed fibroblasts impacts other cells that were not directly exposed to dextrose. To do this, we first exposed MRC-5 fibroblasts to dextrose concentrations (5-25%) for 15 min-2hr, removed the supernatant fluid, and then added fresh media (without dextrose) for 8 hrs. This allowed the media to collect any secreted factors in response to dextrose prolotherapy treatments. After 8hrs, the media from the dextrose-treated fibroblasts was transferred to nascent MRC-5 fibroblasts that had never been exposed to dextrose for 48 hrs. The viability of these nascent fibroblasts was then quantified using an XTT assay.
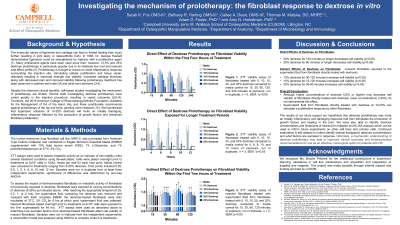
Results: When comparing the viability of fibroblasts directly treated with dextrose solutions, we found that higher concentrations of dextrose solutions may negatively impact fibroblast viability whereas lower concentrations may cause a positive impact on viability compared to media controls. Two-way ANOVA analysis of fibroblasts treated directly with 20% or 25% dextrose for 2hrs or longer significantly reduced cell viability (p < 0.005) compared to the media control. In contrast, preliminary data from nascent fibroblasts (i.e., not directly treated with dextrose) indicates that the secreted factors in the supernatant fluid from dextrose-treated fibroblasts may actually have a positive impact on cell viability.
Conclusion: The results of our study support the hypothesis that dextrose prolotherapy may incite an initially inflammatory and damaging response that then stimulates the production of growth factors and healing in the joint. Further exploration is needed to determine the optimal therapeutic range of dextrose concentrations to use in prolotherapy and what factors are secreted into the joint space to generate this response to dextrose. With this knowledge, hopefully, we can better understand the mechanism by which dextrose prolotherapy promotes the body’s self-healing leading to improved function and reduced pain experienced by patients with osteoarthritis . Ultimately, this understanding may lead to evidence-based recommendations for the use of dextrose prolotherapy as an effective, nonsurgical option that is minimally invasive, inexpensive, and demonstrates few adverse events and less reliance upon medications for pain management. Furthermore, improving stability and structure in patients with ligament and tendon changes will ultimately improve their clinical outcomes and quality of life. We seek to better understand the mechanism of how dextrose prolotherapy works to further contribute to this field of medicine by improving clinical outcomes and providing an efficacious non-surgical treatment option for patients with osteoarthritis.
Acknowledgement of Research Study Sponsors and IRB: The study does not require IRB approval and was possible through internal funding provided by CUSOM.
Methods: The XTT assay was used to determine cell viability on MRC-5 fibroblasts directly treated with a range of dextrose concentrations (5 -25%) in complete media across 15 min-12 hr. Additionally, we investigated how the response of dextrose-exposed fibroblasts impacts other cells that were not directly exposed to dextrose. To do this, we first exposed MRC-5 fibroblasts to dextrose concentrations (5-25%) for 15 min-2hr, removed the supernatant fluid, and then added fresh media (without dextrose) for 8 hrs. This allowed the media to collect any secreted factors in response to dextrose prolotherapy treatments. After 8hrs, the media from the dextrose-treated fibroblasts was transferred to nascent MRC-5 fibroblasts that had never been exposed to dextrose for 48 hrs. The viability of these nascent fibroblasts was then quantified using an XTT assay.
Results: When comparing the viability of fibroblasts directly treated with dextrose solutions, we found that higher concentrations of dextrose solutions may negatively impact fibroblast viability whereas lower concentrations may cause a positive impact on viability compared to media controls. Two-way ANOVA analysis of fibroblasts treated directly with 20% or 25% dextrose for 2hrs or longer significantly reduced cell viability (p < 0.005) compared to the media control. In contrast, preliminary data from nascent fibroblasts (i.e., not directly treated with dextrose) indicates that the secreted factors in the supernatant fluid from dextrose-treated fibroblasts may actually have a positive impact on cell viability.
Conclusion: The results of our study support the hypothesis that dextrose prolotherapy may incite an initially inflammatory and damaging response that then stimulates the production of growth factors and healing in the joint. Further exploration is needed to determine the optimal therapeutic range of dextrose concentrations to use in prolotherapy and what factors are secreted into the joint space to generate this response to dextrose. With this knowledge, hopefully, we can better understand the mechanism by which dextrose prolotherapy promotes the body’s self-healing leading to improved function and reduced pain experienced by patients with osteoarthritis . Ultimately, this understanding may lead to evidence-based recommendations for the use of dextrose prolotherapy as an effective, nonsurgical option that is minimally invasive, inexpensive, and demonstrates few adverse events and less reliance upon medications for pain management. Furthermore, improving stability and structure in patients with ligament and tendon changes will ultimately improve their clinical outcomes and quality of life. We seek to better understand the mechanism of how dextrose prolotherapy works to further contribute to this field of medicine by improving clinical outcomes and providing an efficacious non-surgical treatment option for patients with osteoarthritis.
Acknowledgement of Research Study Sponsors and IRB: The study does not require IRB approval and was possible through internal funding provided by CUSOM.
