Health Policy
Evaluating guidelines for tobacco cessation in pregnancy: an appraisal using the AGREE II instrument
Friday, March 31, 2023
12:00 PM East Coast USA Time

Mohit Chhatpar, DO (he/him/his)
Indiana Regional Medical Center
Indiana, Pennsylvania, United States
Presenting Author(s)
Background and Hypothesis: Smoking is the most important preventable cause of adverse outcomes in pregnancy; however, most smokers who become pregnant continue to smoke and/or relapse after delivery. Clinical practice guidelines (CPGs) have been published to help identify at-risk patients and guide holistic management of tobacco use in pregnancy, but to date, there has been no comprehensive review of guideline quality or methodological rigor. Therefore, this study set out to identify and assess the methodologic quality of national and international guidelines for the evaluation and management of tobacco cessation in pregnancy.
Methods: We conducted a comprehensive search of EMBASE, MEDLINE/PubMed, SCOPUS and grey literature sources until October 2022. The quality of these guidelines was assessed by four reviewers using the Appraisal of Guidelines for Research and Evaluation, 2nd edition (AGREE II). Domain scores were considered acceptable quality if they scored >60%, and Intraclass Correlation Coefficients (ICC) were calculated to assess agreement among the appraisers.
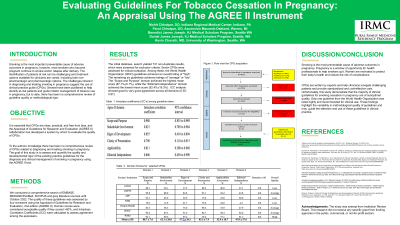
Results: Seven CPGs were assessed for critical evaluation. Among these, the World Health Organization (WHO) guidelines achieved an overall rating of "high”. The remaining six guidelines achieved ratings of “average” or “low”. The “Scope and Purpose” domain achieved the highest mean score (87.1%±0.1%), while the “Editorial Independence” domain achieved the lowest mean score (52.4%±19.3%). ICC analysis showed good to very good agreement across all domains (0.55-0.97).
Conclusion: The overall quality of clinical practice guidelines for the management of smoking cessation in pregnancy are suboptimal, with only one guideline rated highly and recommended for clinical use. These findings highlight the variability in methodologic quality of guidelines and may guide the selection and use of these guidelines in clinical practice.
Acknowledgement of Research Study Sponsors and IRB: The study was exempt from Institution Review Board. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Methods: We conducted a comprehensive search of EMBASE, MEDLINE/PubMed, SCOPUS and grey literature sources until October 2022. The quality of these guidelines was assessed by four reviewers using the Appraisal of Guidelines for Research and Evaluation, 2nd edition (AGREE II). Domain scores were considered acceptable quality if they scored >60%, and Intraclass Correlation Coefficients (ICC) were calculated to assess agreement among the appraisers.
Results: Seven CPGs were assessed for critical evaluation. Among these, the World Health Organization (WHO) guidelines achieved an overall rating of "high”. The remaining six guidelines achieved ratings of “average” or “low”. The “Scope and Purpose” domain achieved the highest mean score (87.1%±0.1%), while the “Editorial Independence” domain achieved the lowest mean score (52.4%±19.3%). ICC analysis showed good to very good agreement across all domains (0.55-0.97).
Conclusion: The overall quality of clinical practice guidelines for the management of smoking cessation in pregnancy are suboptimal, with only one guideline rated highly and recommended for clinical use. These findings highlight the variability in methodologic quality of guidelines and may guide the selection and use of these guidelines in clinical practice.
Acknowledgement of Research Study Sponsors and IRB: The study was exempt from Institution Review Board. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
